Water aka H2O=Polar Molecule
Polarity-ability of water to interact with other molecules and compounds due to its positive H+ charges and negative O- charge.
Properties that H2O can exhibit:
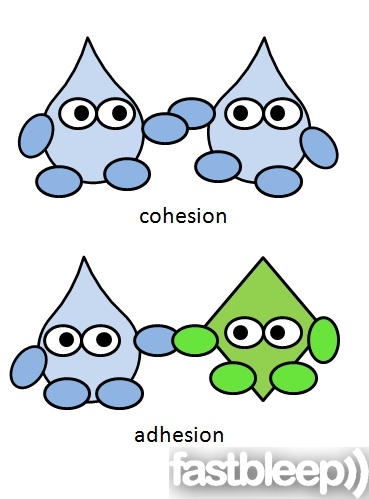
Cohesion-Water's attraction to other water molecules
Adhesion-Water's attraction to other molecules
Surface Tension-the attraction of water molecules along the surface (along bugs to walk across water)
Capillary Action-the ability of water to rise against the force of gravity due to the adhesiveness of water on the sides of the tube
High Specific Heat - water's ability to maintain a constant temperature (such as in oceans); the amount of heat one gram of water must absorb or lose to change its temperature by one degree Celsius.
Cohesion-Water's attraction to other water molecules
Adhesion-Water's attraction to other molecules
Surface Tension-the attraction of water molecules along the surface (along bugs to walk across water)
Capillary Action-the ability of water to rise against the force of gravity due to the adhesiveness of water on the sides of the tube
High Specific Heat - water's ability to maintain a constant temperature (such as in oceans); the amount of heat one gram of water must absorb or lose to change its temperature by one degree Celsius.